 
Since S = 0 corresponds to perfect order. The entropy of a pure crystalline substance at absolute zero (i.e. Nonetheless, the combination of these two ideals constitutes the basis for the third law of thermodynamics: the entropy of any perfectly ordered, crystalline substance at absolute zero is zero. In practice, absolute zero is an ideal temperature that is unobtainable, and a perfect single crystal is also an ideal that cannot be achieved. Such a state of perfect order (or, conversely, zero disorder) corresponds to zero entropy. The only system that meets this criterion is a perfect crystal at a temperature of absolute zero (0 K), in which each component atom, molecule, or ion is fixed in place within a crystal lattice and exhibits no motion (ignoring quantum effects). A perfectly ordered system with only a single microstate available to it would have an entropy of zero. The greater the molecular motion of a system, the greater the number of possible microstates and the higher the entropy. These forms of motion are ways in which the molecule can store energy. due to the reduction in the degrees of freedom, the system is more ordered after the reaction). There is a reduction in the disorder of the system (i.e. 
The reaction has resulted in a loss of freedom of the atoms (O atoms) The 3rd law of thermodynamics says that a perfect (100 pure) crystalline structure at absolute zero (0 K) will have no entropy ((S)).Since they are now physically bonded to the other molecule (forming a new, larger, single molecule) the O atoms have less freedom to move around. 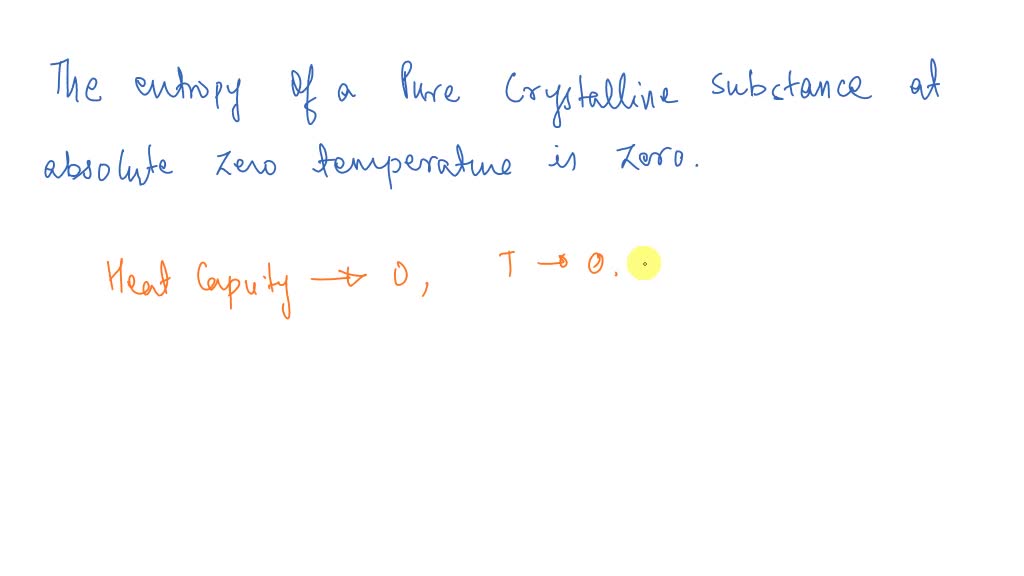
The product of this reaction (\(NO_2\)) involves the formation of a new N-O bond and the O atoms, originally in a separate \(O_2\) molecule, are now connected to the \(NO\) molecule via a new \(N-O\) bond. Nernst’s heat theorem and Planck’s extension of it, while originally derived from observing the behaviour of chemical reactions in solids and liquids, is now believed to apply quite generally to any processes, and, in view of that, it is time to reconsider our description of adiabatic demagnetization. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
